|
5/20/2023 0 Comments Kidney and adrenal gland
It is involved in the discovery, development, production and commercialization of pharmaceuticals and biotechnology products. Kyowa Kirin, a subsidiary of Kirin Holdings Co Ltd, is a biotechnology company focused on research and development. It was also under development for urothelial carcinoma, advanced or metastatic solid tumors including metastatic urothelial tract cancer, bladder cancer, urethral cancer, ureter cancer, renal pelvis cancer. The drug candidate is administered through oral route and formulated as a solid encapsulated dosage form. 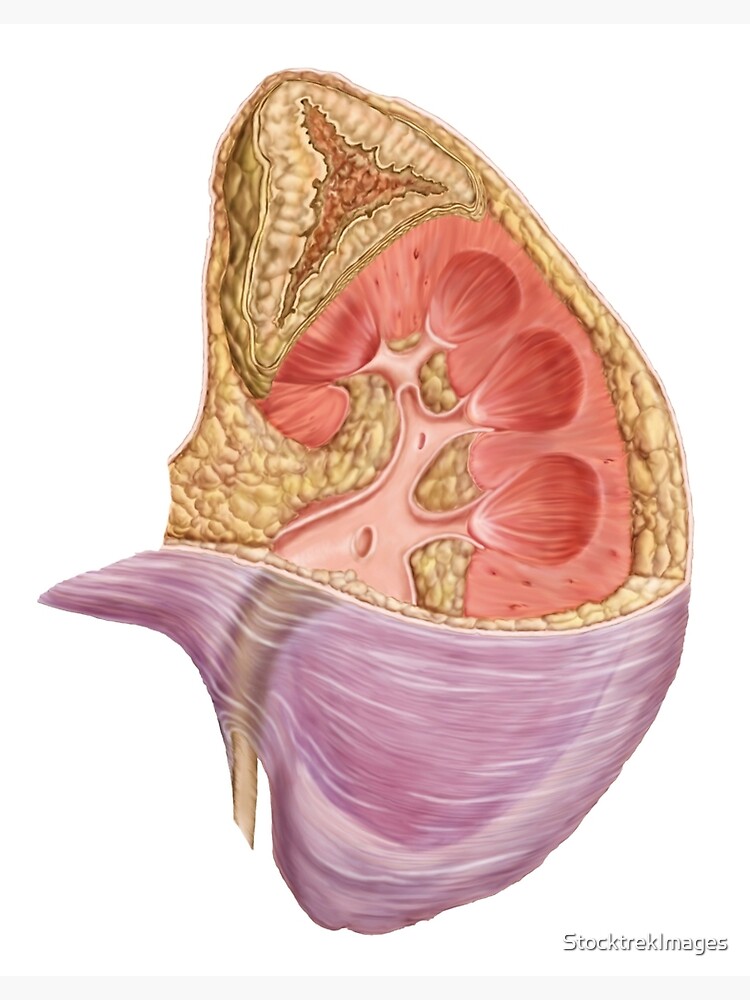
KHK-2455 is under development for the treatment of head and neck cancer, ovarian cancer, osteosarcoma, pancreatic cancer, gastric cancer, gallbladder cancer and adrenal cancers. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data. GlobalData’s report assesses how KHK-2455’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks. According to GlobalData, Phase I drugs for Adrenal Gland Cancer have an 86% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. 
KHK-2455 is under clinical development by Kyowa Kirin and currently in Phase I for Adrenal Gland Cancer.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
